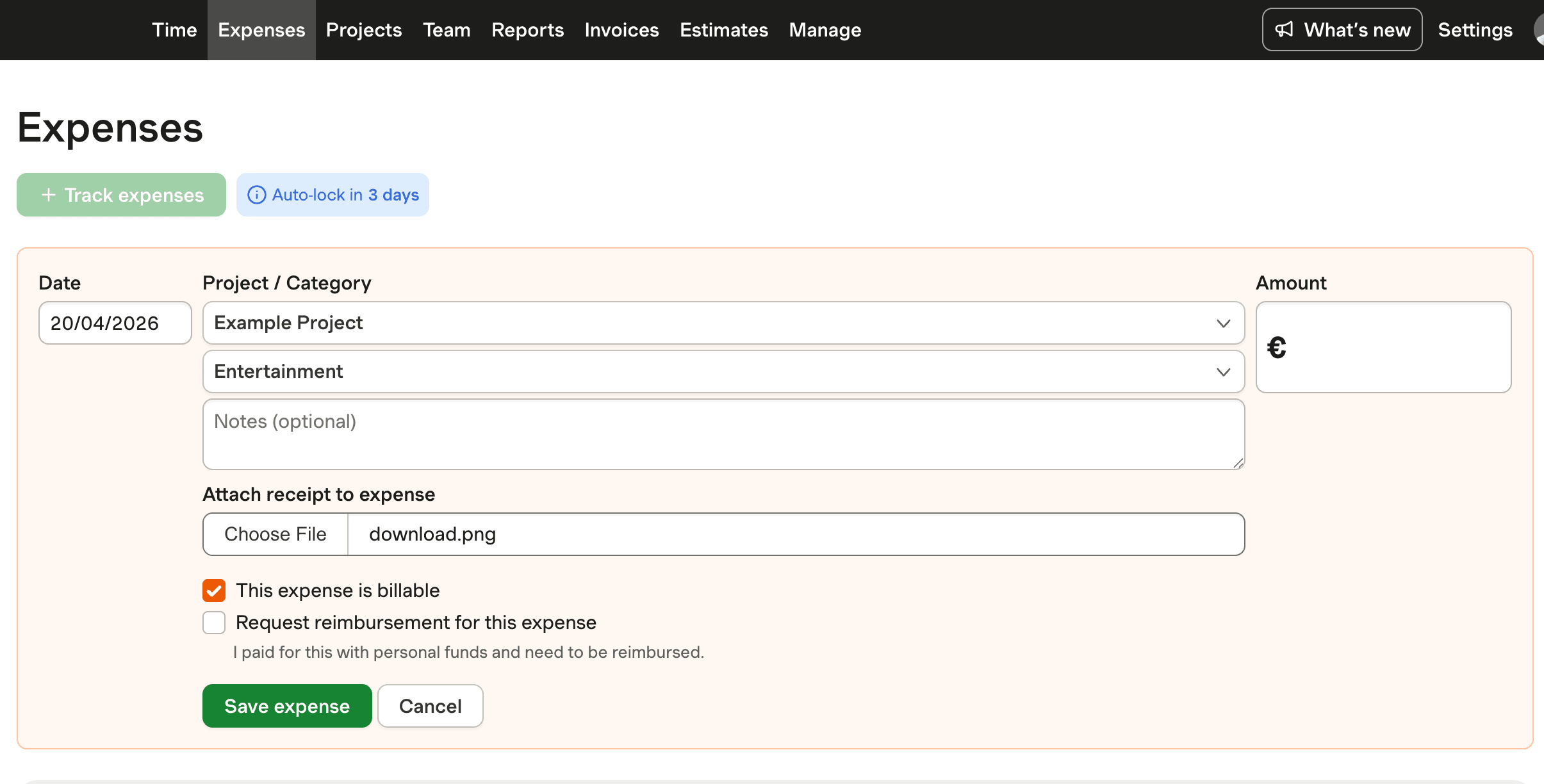
Understanding Pharmaceutical Expense Reporting
Pharmaceutical companies face unique challenges in expense reporting due to strict regulatory requirements and the complex nature of their operations. The industry has seen a significant increase in expenditures, with overall pharmaceutical spending in the US growing by 7.7% in 2021 to reach $576.9 billion. This financial complexity necessitates precise documentation and compliance with regulations like the Sunshine Act and FDA audit mandates. Failure to adhere to these can result in substantial penalties, with companies having paid $62.3 billion in fines over the last three decades due to deficiencies in tracking systems.
Effective expense management is critical, not only for compliance but also for strategic financial planning. Automation of repetitive tasks within expense management can reduce manual effort and processing costs, enabling teams to focus on strategic analysis. Despite these needs, only 10% of pharmaceutical executives have adopted large-scale zero-based budgeting (ZBB) programs, which could deliver up to 25% in cost savings. Understanding these dynamics is essential for creating a robust expense report that meets industry standards.